
The nuclear binding energy varies between nuclei.This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons. The neutron is slightly heavier than the proton.There are two reasons for the difference between mass number and isotopic mass, known as the mass defect: For example, 63Cu (29 protons and 34 neutrons) has a mass number of 63 and an isotopic mass in its nuclear ground state is 62.91367 u. For other isotopes, the isotopic mass usually differs and is usually within 0.1 u of the mass number. One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C the atomic mass is exactly 12u, since the atomic mass unit is defined from it. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). Units of measure have been defined for mass and energy on the atomic scale to make measurements more convenient to express. The size and mass of atoms are so small that the use of normal measuring units, while possible, is often inconvenient. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Because it’s a liquid and not a solid, it can be hard to work in many places and can be hard to keep under control.The atomic mass is the mass of an atom. Liquid nitrogen is much colder than dry ice, which makes it more dangerous to handle. Liquid nitrogen (LN) is an inert cryogenic fluid with a temperature of − 196 ☌. It is supplied as a compressed, liquefied gas. It is also used in the manufacturing of pharmaceuticals, disinfectants, and other chemicals. It is used as a reducing agent and an analytical reagent. Aqueous solutions of HI are known as hydroiodic acid or hydriodic acid, a strong acid. Hydrogen iodide (HI) is a diatomic molecule and hydrogen halide. This means it can be used as a fluorine source. Unlike fluorinated carbons, nitrogen trifluoride is easily degraded due to the low bond energy in the N–F bond. It is primarily used in the production of semiconductors and LCD (Liquid Crystal Display) panels, as well as some types of solar panels and chemical lasers, but not all of them. It is used in the electronics industry and in high-power lasers because it is a source of fluorine. Nitrogen trifluoride is an odorless, colorless gas. Hydrogen gas is a loose aggregation of hydrogen molecules, each of which has two atoms, making it a diatomic molecule, H2. 
The nucleus of a hydrogen atom is made up of one proton with one unit of positive electrical charge and one electron with one unit of negative electrical charge. Hydrogen (H) is the most fundamental member of the chemical element family and is a colorless, odorless, tasteless, flammable gaseous substance. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75 percent of all normal matter. It is colorless, odorless, tasteless, non-toxic, and highly combustible. 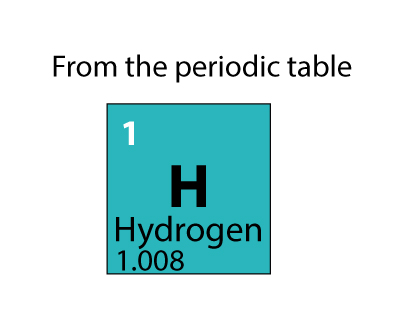
At standard conditions, hydrogen is a gas of diatomic molecules having the formula H 2. Hydrogen is the chemical element with the symbol H and atomic number 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
